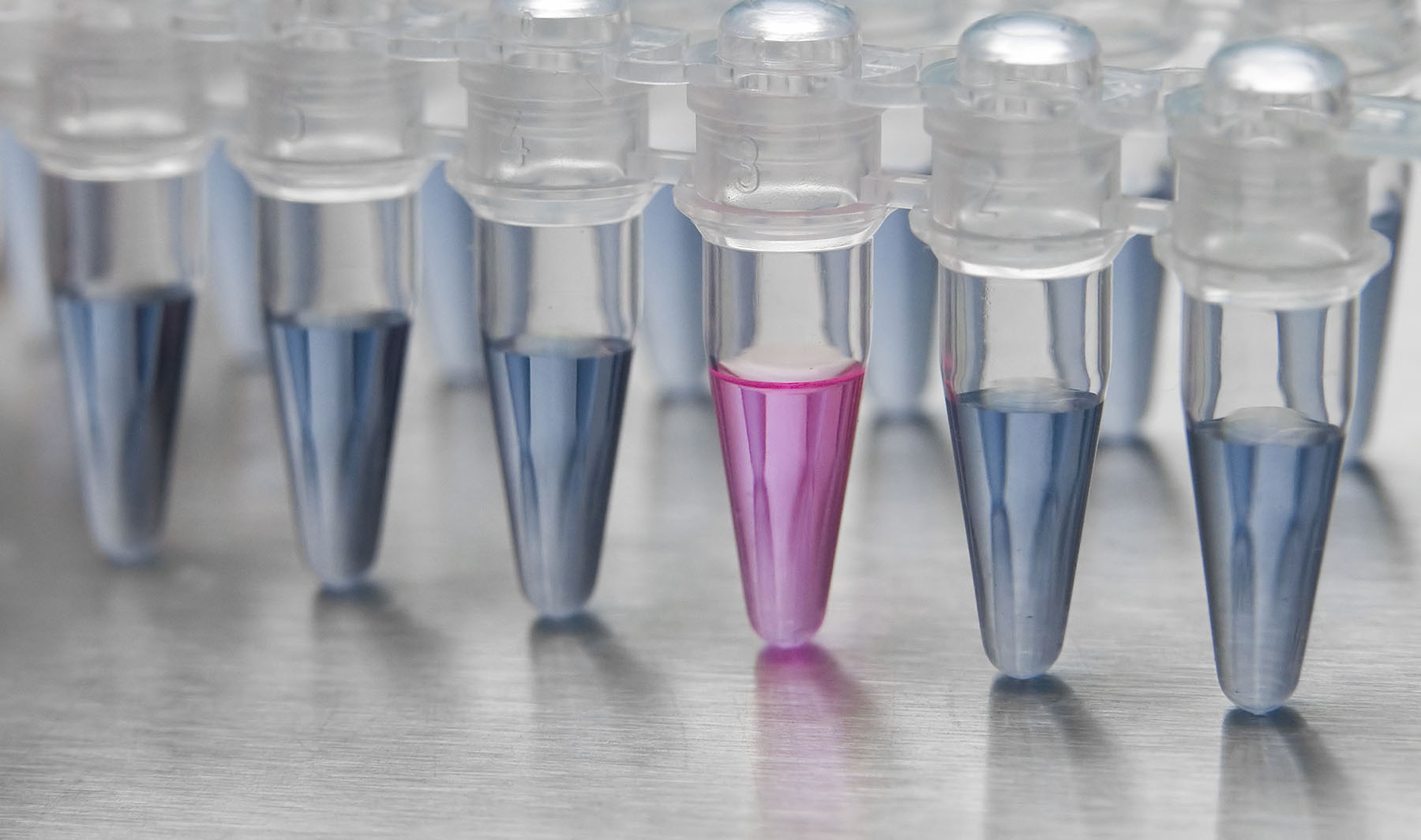
Ensure Quality Products with a Trusted Partner
The global pharmaceutical market has surpassed one trillion dollars annually, and manufacturers are working with only their most trusted partners to produce quality products. For more than 50 years, MRIGlobal has been that partner, providing clinical research support to government agencies and commercial clients. Today, our capabilities span most of the drug development pipeline, from pre-clinical studies to commercialized pharmaceuticals.
Two of our laboratories are proudly named the “Tom Sack Laboratories,” dedicated to our former President & CEO. Here, we conduct GMP pharmaceutical analysis work, supporting human clinical trials. This includes sample preparation, analytical method development and validation, as well as stability testing to establish the shelf-life of drug products and active pharmaceutical ingredients.
Our world-class instrumentation, flexibility to customize solutions, and high standards for execution all help ensure the quality of your products, building your share in this global market.
GETTING STARTED WITH MRIGLOBAL
Contact MRIGlobal to further understand our work in pharmaceutical sciences. We help clients develop faster bench-to-bedside processes to deliver therapeutic drugs to clinics for efficacy testing by bringing unique solutions to pharma from drug discovery to manufacturing.
To learn more about the work we’ve done or how we can help you, contact us today. If you are part of an agency, business, or academic institution seeking assistance with a project, use our Project Quote Tool to get started.
SIGN UP FOR OUR NEWSLETTER
Sign up for the MRIGlobal newsletter! It’s the best way to get the latest updates in the world of applied scientific and engineering research delivered directly to your inbox.